Source: What is Pure Substance? – Definition, Examples, Difference between Pure Substance & Mixture (byjus.com)
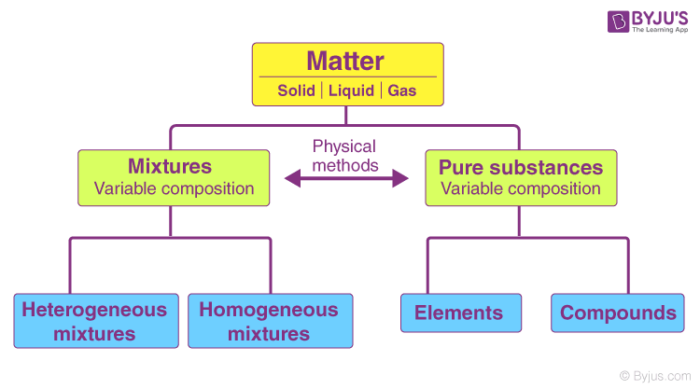
Everything that exists in the earth is a form of a matter which is further defined as any substance that occupies space and has mass. The matter is further divided into various forms such as solid, liquid and gas. Apart from these, it is also classified as pure substances and mixtures.
Quick definitions
- Pure substances: are substances that are made up of only one type of matter and has constant properties throughout the substance.
- Impure substances: are made up of 2 or more types of matter and can have different properties throughout the substance. (These are more commonly know as Mixtures, which are further defined below)
- Homogeneous Mixture: occasionally called a solution, is comparatively unvarying in configuration or constant. Every unit of the mixture is like every other unit. (For instance, if you liquefy sugar in water and blend it really well, your solution is essentially the same, no matter where you sample it. This mixture contains two or more chemical substances.)
- Heterogeneous Mixture: is a concoction whose configuration varies from spot to spot within the sample. (For example, if you put a little amount of sugar in a vessel, add some sand, and then shake the jar a couple of times, your concoction doesn’t have the same configuration all throughout the jar. As the sand is heftier, there’s possibly more amount of sand at the bottom of the jar and more sugar at the top part. These mixtures can be identified visually and separated easily by physical means.)
Differences Between Pure Substances and Mixtures
| Pure Substances | Mixtures |
| It cannot be broken down or separated into new products. | It can be separated using different separation methods. |
| Constant physical and chemical properties. | Mixtures have varying physical and chemical properties. |
| Pure substances are made up of a single element. | A mixture is a combination of two substances or elements. |
What are some examples of Pure Substances and Heterogenous and Homogenous Mixtures?
Make 3 columns on your page with the 3 headings, brainstorm as a class what matter goes into which column.